 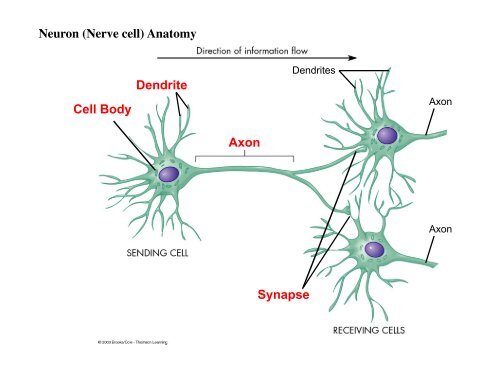
The considerable significance of the genetics associated with autism has led to the identification of many risk genes for ASD used for the probing of ASD specificity and shared cognitive features over the past few decades. doi: 10.1523/ spectrum disorder (ASD) is a genetic heterogeneous neurodevelopmental disorder that is characterized by impairments in social interaction and speech development and is accompanied by stereotypical behaviors such as body rocking, hand flapping, spinning objects, sniffing and restricted behaviors. Na v1.1 localizes to axons of parvalbumin-positive inhibitory interneurons: A circuit basis for epileptic seizures in mice carrying an Scn1a gene mutation. Ogiwara, I., Miyamoto, H., Morita, N., Atapour, N., Mazaki, E., Inoue, I., Takeuchi, T., Itohara, S., Yanagawa, Y., Obata, K., Furuichi, T., Hensch, T.K. “We hope to develop effective therapies for this intractable epilepsy from further work,” says project leader Kazuhiro Yamakawa. In particular, they are found on inhibitory nerve cells that express the calcium-binding protein parvalbumin, often in the area known as the axon initial segment where nerve impulses are generated.īy measuring and comparing the output of excitatory and inhibitory neurons in normal and mutant mice, the research team found that the Na v1.1 channel proteins were needed not to initiate firing of the excitatory nerve, but to maintain the inhibitory pulse, thus preventing epileptic seizures. 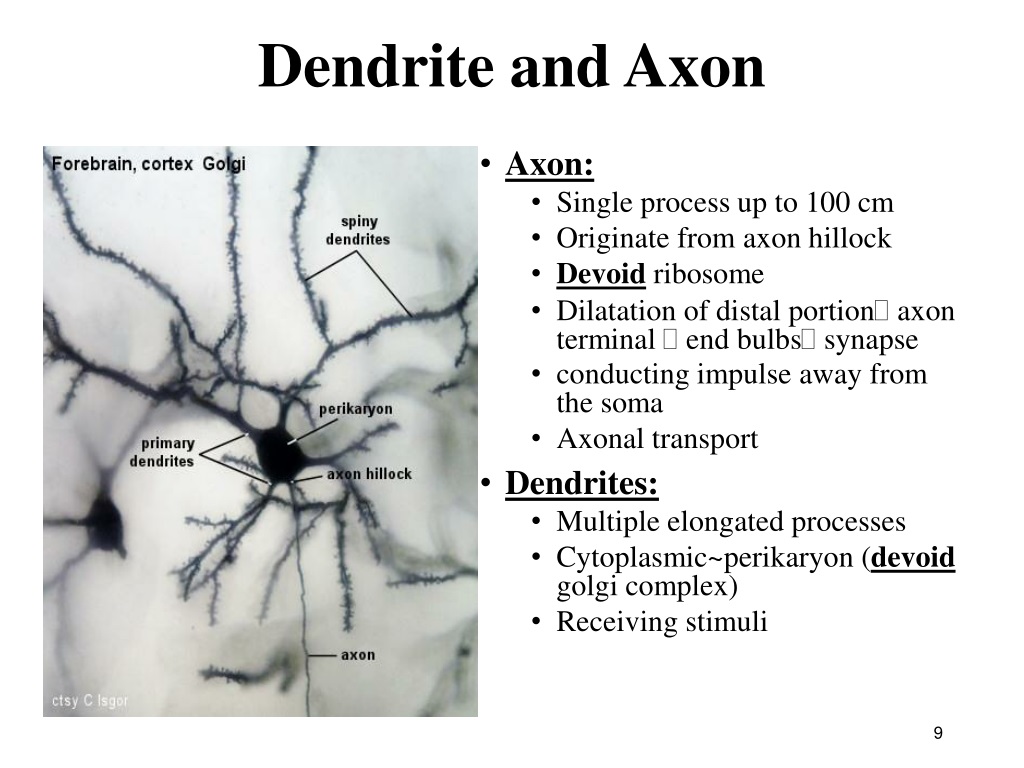
The Na v1.1 proteins are more likely to be found on axons and cell bodies. Using three different antibodies as probes, the RIKEN-based research team corrected this picture. Previous studies suggested that the Na v1.1 protein was distributed rather evenly throughout the brain and could be found in the projections of nerve cells known as dendrites. Pups with only one copy of the mutant gene began epileptic seizures in the third week, and about 40% had died within three months. 1), developed epilepsy and an unstable gait by the second week after birth, and died within three weeks. Mouse pups which inherited copies of the mutant gene from both mother and father were markedly smaller (Fig. In a recent paper in The Journal of Neuroscience 1, researchers from the RIKEN Brain Science Institute, Wako, and their colleagues, describe how they produced a ‘knock-in’ mouse, by introducing a disease-causing, nonsense mutation found in SMEI patients into the middle of the Scn1a gene. There is a homologous gene, Scn1a, in mice. This is a significant step in the generation of nerve impulses. The gene itself encodes an ion-channel protein, Na v1.1, which forms a pore in the plasma membrane that controls the in-flow of electrically-charged sodium ions into nerve cells. More than 200 different mutations of the human SCN1A gene are known to be associated with human epileptic disorders including SMEI. The work also has generated a mouse model of severe myoclonic epilepsy that the researchers hope to use to study the condition and how to treat it. In the process they have redefined the position and role of an important protein involved in controlling the firing of nerve impulses in the brain. Japanese neuroscientists have clarified the molecular basis of the intractable epileptic disorder known as severe myoclonic epilepsy in infancy (SMEI). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
